MDCG 2019-14 Rev.0
Explanatory note on MDR codes
Disclaimer: This document is an interactive version of the original MDCG document. We will keep it up-to-date.
This document has been endorsed by the Medical Device Coordination Group (MDCG) established by Article 103 of Regulation (EU) 2017/745. The MDCG is composed of representatives of all Member States and it is chaired by a representative of the European Commission.
MDCG 2020-10/1 Rev.1 changes
Insert Table of changes here.
1. Introduction
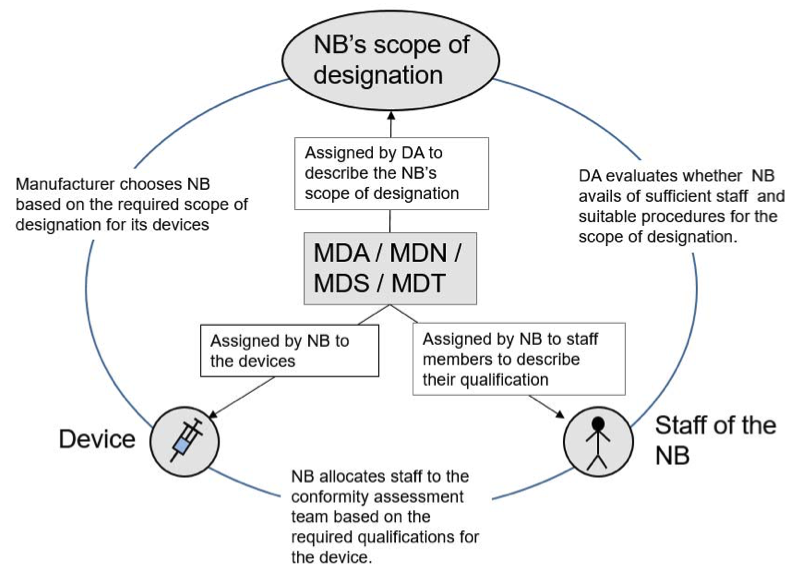
Commission Implementing Regulation 2017/2185 establishes the codes for the designation of notified bodies in medical devices under Regulation (EU) 2017/745 and in vitro diagnostic medical devices under Regulation (EU) 2017/746. These codes are primarily used by designating authorities to define the notified body scope of designation but they are also used by the notified body to:
1) describe the individual qualification of the NBs staff members
2) describe the qualification required for assessing a device
These codes may be very broad and, furthermore, unequivocal authorisation of personnel to codes and the assignment of codes to a device is not always straightforward. However, the notified body’s system needs to ensure, in all cases, that the authorisation of personnel and team allocation for the conformity assessment of a device ensures adequate knowledge and expertise.
2. Scope
The lists of codes and corresponding types of devices established by the above mentioned Regulation takes into account various device types which can be characterised by design and intended purpose, manufacturing processes and technologies used, such as sterilisation and the use of nanomaterials.
These lists of codes should be used in a way that provides for a multi-dimensional application to all typology of devices. This will ensure that notified bodies as well as the staff assigned to conformity assessment are fully competent for the devices they are required to assess.
This guidance is intended to explain the different level of codes and how they should be used, including the use of conditions with a view to ensure a harmonised use of the codes especially for the allocation of resources to conformity assessment activities.
3. Assignment of codes to devices within the conformity assessment procedure
When a manufacturer lodges an application with a notified body, the type of devices and technologies subject to conformity assessment activities are to be indicated. Usually, at the application review stage (as defined in section 4.3 of Annex VII MDR), notified bodies will verify the assignment of codes provided by the manufacturer or will assign these codes to the devices themselves. This verification is carried out in order to ensure that the notified body is able to assess the application based on its designation, and that it has available resources to carry out the relevant conformity assessment activities (feasibility evaluation). Thefinal assignment is made by the NB.
After this application review, and signing of the contract, the notified body will allocate appropriately qualified and authorised personnel to carry out audit activities or product reviews.
The following table presents an overview of the different types of codes and a summary of the main characteristics of each of them for the assignment to specific devices and the allocation of resources.
Type Code | Assignment of codes to the device | Relevance for allocation of conformity assessment team |
|---|---|---|
MDA / MDN | Exactly 1 code per device. The codes should be selected according to their hierarchical order in Regulation 2017/2185. If more than 1 MDA/ MDN code is applicable, the one that is highest in the list should be selected. | Allocation of personnel involved in the review of technical documentation (e.g. product reviewers) or in audits concerning product related aspects. |
MDS | 0 to several per device Assign all codes applicable to the device. Select once an MDN/MDA code has been assigned. | Allocation of personnel involved in the review of technical documentation (e.g. product reviewers of sterilisation validation). May also be applicable to staff performing audits concerning certain special processes (e.g. sterilization).* |
MDT | 1 to several per device Assign the codes which Select once an MDN/MDA code has been assigned. | Allocation of personnel involved in audits (e.g. site auditors involved in the auditing of metal processing). |
* Note: Assessment of these codes could be performed by Product reviewers or Site auditors depending on their competence
3.1 MDN / MDA-codes
MDA / MDN-codes reflect the design and intended purpose of the device and hence are mostly relevant for the allocation of personnel involved in the review of technical documentation. In some specific cases, the NB may assign product reviewers to assess product performance and safety aspects during an audit. This means that if there are product related issues to be audited and the auditors do not possess the required qualification, product reviewers who are qualified for the device in question should be part of the audit team.
The NB needs to ensure that the personnel allocated to the project are competent to assess for the devices and technologies under assessment. Special attention should be paid in situations such as the one described in example 4 of this section.
The MDA / MDN codes may either specify a field of medical application (e.g. MDA 0309 Active non- implantable ophthalmologic devices) or the physical or technological principle of the device (e.g. MDA 0302 Active non-implantable devices utilising non-ionizing radiation or MDA 0315 Software). Therefore, there are cases where more than one specific code might apply to a device (e.g. surgical laser for refractive surgery of the eye). Also, where there is a broad intended purpose, several codes may apply.
Having these issues in mind, when drafting the Regulation 2017/2185, the codes were put in order such that the MDA/MDN codes that require very specific technological knowledge and experience are highest in the code lists. Therefore, in cases where more than one MDA/MDN code apply, the code highest in the list is to be selected. This approach ensures consistent assignment of codes (and therefore consistent assignment of suitably qualified staff) to devices.
Example 1: A surgical laser for refractive surgery of the eye is assigned to MDA 0302 Active non-implantable devices utilising non-ionizing radiation and not to MDA 0309 Active non- implantable ophthalmologic devices because, even though both codes are specific for the device, since MDA 0302 is higher in the list.
Example 2: A screw for orthopaedic surgery is assigned to MDN 1102 Non-active osteo- and orthopaedic implants and not to 1104 Non-active soft tissue and other implants, because MDN 1102 is higher in the list.
Devices may be composed of different “components” which, if they were products on their own, would belong to different MDA/MDN codes (1). In such cases, the intended purpose or the main physical or technological principle of the device should be considered (Example 3). Nonetheless, the notified body needs to ensure that the assigned staff is qualified to assess all components of the device.
Example 3: A medical devices is composed of a suture anchor (a bone screw attached to a surgical suture to reattach ruptured tendons) as well as a single use deployment instrument and a single use bone drill. The components are provided sterile in a blister, are covered by the same technical documentation and are not available individually (e.g. are not medical devices on their own). This product is assigned to MDN 1102 Non-active osteo- and orthopaedic implants and not to MDN 1208 Non-active non-implantable instruments because the implanted component of the device is associated with the intended purpose rather than the deployment instrument and the drill.
Note that, given the complexity and the diversity of medical devices, in exceptional cases deviations from the guidance given above may be necessary when assigning codes to devices in order to ensure suitably qualified staff in the conformity assessment. In such cases, a brief rationale shall be documented (see Example 4).
Example 4: A heater-cooler unit (HCU) for cardiac surgery is a device through which blood circulates, and which changes the circulating blood’s temperature in order to achieve hypo- or hyperthermia. Both the codes MDA 0303 Active non-implantable devices utilising hyperthermia/hypothermia and MDA 0306 Active non-implantable devices for extra-corporal circulation, administration or removal of substances and haemapheresis are specific to the device. Since MDA 0303 Active non-implantable devices utilising hyperthermia/hypothermia is a code that describes physical or technological characteristics higher in the list, according to the rule explained above MDA 0303 should be chosen. Nonetheless, the notified body assigns the device to the code MDA 0306 Active non-implantable devices for extra-corporal circulation, administration or removal of substances and haemapheresis and documents that the risks associated with the HCU are such that the staff having experience in this code is more suitable to assess the device.
It is important to note that Stand-alone software (i.e. software that is not part of a physical medical device) should be assigned to MDA 0315 since assessment of software requires very specific knowledge (see annex VII 3.2.2 5th indent MDR) and also that non-implantable cardiovascular catheters, guidewires, introducers, filters and related tools shall fall under MDN 1203 (instead of MDN 1201 or MDN 1202).
3.2 MDS codes
MDS codes are horizontal codes that are applicable to devices with specific characteristics. All codes that are applicable need to be assigned to a device in order to ensure that the review team possesses the full set of qualifications necessary for the conformity assessment. The MDS-codes are mainly relevant for the allocation of personnel involved in the review of technical documentation. This is because, generally, the auditing aspects linked to MDS codes have their corresponding MDT code for the relevant technology (e.g. MDS 1001 Devices incorporating medicinal substances vs MDT 2007 Devices which require knowledge regarding the production of pharmaceuticals). However, MDS codes may also be applicable to staff performing audits concerning certain special processes (for example MDS 1005 for staff auditing ethylene oxide sterilization processes).
Example 1: A partially resorbable, sterile surgical implant that contains an antibiotic to prevent post-surgical infection will need to be assigned to the following MDS codes:
1) MDS 1005 – Devices in sterile condition: because it is provided sterile,
2) MDS 1008 – Devices utilising […] being wholly or mainly absorbed or locally dispersed in the human body […]: because it is absorbed, and
3) MDS 1001 – Devices incorporating medicinal substances: because it contains an antibiotic.
Example 2: An infusion pump should be assigned to MDS 1009 – Devices incorporating software/utilising software/controlled by software […] because the infusion pump is controlled by software.
3.3 MDT codes
MDT codes relate to the technologies and processes that are used in the manufacturing and making available of the devices. MDT codes are relevant for the allocation of site auditors.
Assignment of MDT codes should be done taking into consideration production of the device itself as well as for critical upstream production steps. This means that, even though many codes could be applicable when taking into consideration the processes involved in the entire supply chain of a medical device, these should not be considered for the use of MDT codes (e.g. for an electronic medical thermometer, at one point, metal processing, plastic processing, non-metal mineral processing, chemical processing, manufacture in clean rooms, manufacturing using electronic components, labelling and packaging are necessary to assemble the device from raw materials).
Example 1: An electronic medical thermometer for layman’s use should be assigned to the following MDT codes:
1) MDT 2010 Devices manufactured using electronic components including communication devices because the product is assembled from electronic components and
2) MDT 2011 Devices which require packaging, including labelling because the device is packed and labelled.
Example 2: A sterile animal derived bone graft substitute is manufactured using several steps. In the first step, the raw animal bone undergoes a sequence of chemical treatments to remove organic components. In a second step, the remaining mineral bone component is subjected to a heat treatment, then ground and sieved to obtain particles of defined sizes. The material is packed, labelled and sterilized. Therefore the following MDT codes should be assigned:
1) MDT 2008 Devices manufactured in clean rooms and associated controlled environments, because the manufacturing of the implant after the heat treatment is conducted in clean rooms,
2) MDT 2009 Devices manufactured using processing of materials of human, animal, or microbial origin because the implant is made from animal bone
3) MDT 2011 Devices which require packaging, including labelling because the implant is packed and labelled.
Note: MDT 2006 Devices manufactured using chemical processing and MDT 2003 Devices manufactured using non-metal mineral processing (e.g. glass, ceramics) were not applied since the chemical treatment and (bone) mineral processing are already considered within the scope of processing of animal materials.
Example 3: A calcium phosphate bone cement is provided as a sterile powder composed of a mixture of calcium salts and a vial with sterile saline. The following MDT codes should be assigned:
1) MDT 2008 Devices manufactured in clean rooms and associated controlled environments, because the manufacturing of the implant is in a controlled environment,
2) MDT 2006 Devices manufactured using chemical processing, because the main risks in manufacturing are related to the testing and mixing of the components,
3) MDT 2011 Devices which require packaging, including labelling because the implant is packed and labelled.
Example 4: A manufacturer of cross-linked hyaluronic acid implants has no in-house manufacturing. The finished, labelled implant is bought from a supplier. The implant raw material (hyaluronic acid) is produced by fermentation using bacteria. The following MDT codes should be assigned:
1) MDT 2008 Devices manufactured in clean rooms and associated controlled environments, because the manufacturing of the implant is in a controlled environment,
2) MDT 2005 Devices manufactured using biotechnology, because the main risks in manufacturing are related to the production and cross-linking of the hyaluronic acid.
3) MDT 2011 Devices which require packaging, including labelling because the implant is packed and labelled.
Note that, despite the fact that the manufacturer itself does not physically manufacture the device, the MDT codes concerning the manufacturing steps are assigned to the device since they are relevant when auditing suppliers / subcontractors.
4. Competence description: conditions (including limitations)
Conditions should be established by the notified body for individual codes in cases where the qualification of the staff authorised to a certain code is not sufficient to cover the entire spectrum of the devices within this code. The designating authority could also apply conditions to the notified body’s designation where the notified body does not have sufficient competence to cover a given code or could seek designation for conformity assessment of only certain devices within a code.
Conditions, including limitations, should be formulated in an unambiguous way (see example 1). Furthermore, since the technical codes basically mirror the competence system of the notified body, the conditions and limitations should concern device characteristics (see example 2).
Example 1: “MDN 1101 Non-active cardiovascular, vascular and neurovascular implants. Condition: heart valves”. This condition is unsuitable since it is not clear whether the notified body is restricted to assessment of heart valves or whether those products are excluded from the scope of designation. Therefore, the condition should be “excluding heart valves” or “including only heart valves”.
Example 2: “MDN 1102 Non-active osteo- and orthopaedic implants. Condition: excluding class III devices”. The scope of designation is intimately linked to the qualification of the staff that the notified body needs to have available. Conditions serve to exclude the devices for which the notified body does not have competent staff and/or suitable procedures; therefore, they need to be expressed in a way that they relate to knowledge and experience of staff and/or procedures. The condition “excluding class III devices” does not allow this and therefore it is unsuitable. In this case, the designating authority has to determine, based on the qualification of the notified body’s staff, which devices are included in the scope. This might result, for example, in the following condition: “including only orthopaedic plates and screws; excluding implants that are applied in or on the spine”.
Further examples of conditions are illustrated in the final column of the table below. Considerations of the conditions should be based on the demonstrated competence of the applicant body.
5. Specific clarifications linked to individual codes
The devices included in the third and fourth columns are only a few examples of the devices covered in the relevant code. The third column is not intended to provide an exhaustive list of devices included in each code.
| MDA CODE | Active implantable devices | Devices covered and Specific Considerations (2) | Examples of conditions |
|---|---|---|---|
| MDA 0101 | Active implantable devices for stimulation / inhibition / monitoring | Implanted defibrillator | Excluding brain stimulation |
| MDA 0102 | Active implantable devices delivering drugs or other substances | Implanted drug delivery pump | |
| MDA 0103 | Active implantable devices supporting or replacing organ functions | Artificial heart | |
| MDA 0104 | Active implantable devices utilising radiation and other active implantable devices | Prostate radioactive seed implant |
MDA CODE | Active non-implantable devices for imaging, monitoring and / or diagnosis | Devices covered and Specific Considerations | Examples of conditions |
|---|---|---|---|
| MDA 0201 | Active non-implantable imaging devices utilising ionizing radiation | X-ray computer tomography equipment | Restricted to X-ray and gamma cameras |
| MDA 0202 | Active non-implantable imaging devices utilising non-ionizing radiation | MRI computer tomograph | |
| MDA 0203 | Active non-implantable devices for monitoring of vital physiological parameters | Blood pressure monitor | |
| MDA 0204 | Other active non-implantable devices for monitoring and / or diagnosis | Ocular tonometer |
MDA CODE | Active non-implantable therapeutic devices and general active non-implantable devices | Devices covered and Specific Considerations | Examples of conditions |
|---|---|---|---|
| MDA 0301 | Active non-implantable devices utilising ionizing radiation | Radioactive sources for cancer after loading therapy | |
| MDA 0302 | Active non-implantable devices utilising non-ionizing radiation | Surgical laser for refractive surgery of the eye | Including only microwave and magnetotherapy medical devices |
| MDA 0303 | Active non-implantable devices utilising hyperthermia / hypothermia | Medical heating blanket Warming and cooling blankets | |
| MDA 0304 | Active non-implantable devices for shock-wave therapy (lithotripsy) | Extracorporeal shock wave lithotripsy device | |
| MDA 0305 | Active non-implantable devices for stimulation or inhibition | Automated external defibrillator | |
| MDA 0306 | Active non-implantable devices for extra-corporal circulation, administration or removal of substances and haemapheresis | Haemodialysis machine and equipment | |
| MDA 0307 | Active non-implantable respiratory devices | Medical ventilator | |
| MDA 0308 | Active non-implantable devices for wound and skin care | Water jet for wound debridement | |
| MDA 0309 | Active non-implantable ophthalmologic devices | Aspiration pump for opthalmological use (removal of crystalline residue) | |
| MDA 0310 | Active non-implantable devices for ear, nose and throat | Hearing aids | |
| MDA 0311 | Active non-implantable dental devices | Powered dental surgical unit and hand pieces | |
| MDA 0312 | Other active non-implantable surgical devices | RF electrosurgical generator | |
| MDA 0313 | Active non-implantable prostheses, devices for rehabilitation and devices for patient positioning and transport | Hospital beds | |
| MDA 0314 | Active non-implantable devices for processing and preservation of human cells, tissues or organs including in vitro fertilisation (IVF) and assisted reproductive technologies (ART) | IVF cryopreservation systems blood bank refrigerator | |
| MDA 0315 | Software | Radiotherapy planning system. | |
| MDA 0316 | Medical gas supply systems and parts thereof | Medical gas supply system in a hospital | |
| MDA 0317 | Active non-implantable devices for cleaning, disinfection and sterilisation | Sterilizers like autoclaves etc | |
| MDA 0318 | Other active non-implantable devices |
| MDN CODE | Non-active implants and long term surgically invasive devices | Devices covered and Specific Considerations | Examples of conditions |
|---|---|---|---|
| MDN 1101 | Non-active cardiovascular, vascular and neurovascular implants | Cardiac vascular stent | Including only cardiac stents |
| MDN 1102 | Non-active osteo- and orthopaedic implants | Artificial spinal disc For antibiotic bone cements- MDS 1001 should be applied For absorbable substances MDS 1008 should be applied | |
| MDN 1103 | Non-active dental implants and dental materials | Dental implant | |
| MDN 1104 | Non-active soft tissue and other implants | Hyaluronic acid dermal fillers Bariatric surgery devices: Intragastric Balloons, Gastric Bands, Anti-Reflux implants. For contraceptive intrauterine devices containing copper or silver – MDS 1001 should be applied | Excluding breast implants |
| MDN CODE | Non-active non-implantable devices | Devices covered and specific conditions | Examples of conditions |
|---|---|---|---|
| MDN 1201 | Non-active non-implantable devices for anaesthesia, emergency and intensive care | Device for pleural drainage Masks, tubes for the administration of gases. Endotracheal tube Tracheostomy tubes Endotracheal tube introducers | |
| MDN 1202 | Non-active non-implantable devices for administration, channelling and removal of substances, including devices for dialysis | Intravenous line Hypodermic needle Syringe tubing, bags for injection or transfusion Dialysis filter Dialysis solution Epidural catheters, Urinary Catheter Amnioscopic needles | |
| MDN 1203 | Non-active non-implantable guide catheters, balloon catheters, guidewires, introducers, filters, and related tools | Vascular filter catheter Embolectomy catheter Neurovascular catheters | |
| MDN 1204 | Non-active non-implantable devices for wound and skin care | Wound dressing Cotton wool Surgical gloves For dressings incorporating an antimicrobial agent – MDS 1001 should be applied | |
| MDN 1205 | Non-active non-implantable orthopaedic and rehabilitation devices | Orthoses Crutches Wheelchairs | |
| MDN 1206 | Non-active non-implantable ophthalmologic devices | Contact lenses Eye drops Instrument for ophthalmologic surgery Solutions for contact lenses. | |
| MDN 1207 | Non-active non-implantable diagnostic devices | Thermal expansion thermometer | |
| MDN 1208 | Non-active non-implantable instruments | Instruments for general use in surgery: | |
| MDN 1209 | Non-active non-implantable dental materials | Etching solution for dental use Dental cements (when not considered long term surgically invasive) Dental impression materials | |
| MDN 1210 | Non-active non-implantable devices used for contraception or prevention of the transmission of sexually transmitted diseases | Condom Contraceptive diaphragms | |
| MDN 1211 | Non-active non-implantable devices for disinfecting, cleaning and rinsing | Solutions for disinfecting medical devices | |
| MDN 1212 | Non-active non-implantable devices for processing and preservation of human cells, tissue or organs including in vitro fertilisation (IVF) and assisted reproductive technologies (ART) | Freezing solution for egg cells Embryo transfer catheters Artificial insemination probes Media, substances or mixture of substances intended for washing, separating, sperm immobilizing, cryoprotecting solutions. | |
| MDN 1213 | Non-active non-implantable devices composed of substances to be introduced into the human body via a body orifice or the dermal route | Inhalt Zeile 2 / Spalte 3 | |
| MDN 1214 | General non-active non-implantable devices used in health care and other non- active non-implantable devices | Ultrasound gels |
| MDS CODE | Devices with specific characteristics | Devices covered and specific conditions | Examples of conditions |
|---|---|---|---|
| MDS 1001 | Devices incorporating medicinal substances | Devices incorporating medicinal products including herbal substances and human blood derivatives are included in this code. | |
| MDS 1002 | Devices manufactured utilising tissues or cells of human origin, or their derivatives | Section 5.3.1 of Annex IX applies | |
| MDS 1003 | Devices manufactured utilising tissues or cells of animal origin, or their derivatives | Section 5.3.2 of Annex IX applies Devices manufactured utilising tissues or cells of animal origin, or their derivatives, which are non- viable or rendered non- viable, unless such devices are intended to come into contact with intact skin only | |
| MDS 1004 | Devices which are also machinery as defined in point (a) of the second paragraph of Article 2 of Directive 2006/42/EC of the European Parliament and of the Council (3) | ||
| MDS 1005 | Devices in sterile condition | Including aseptic processing, ethylene oxide gas sterilisation (EOG), low temperature steam, formaldehyde sterilisation, moist heat sterilisation, radiation (gamma, x-ray, electron beam), sterilisation with hydrogen peroxide, sterilisation with liquid chemical sterilising agents, thermic sterilisation with dry heat | |
| MDS 1006 | Reusable surgical instruments | Reusable surgical instruments according to art. 52 (7) c) MDR | |
| MDS 1007 | Devices incorporating or consisting of nanomaterial | ||
| MDS 1008 | Devices utilising biologically active coatings and / or materials or being wholly or mainly absorbed or locally dispersed in the human body or are intended to undergo a chemical change in the body | Section 5.4 of Annex IX applies for devices intended to be introduced into the human body via a body orifice or applied to the skin and devices at are systemically absorbed by the human body in order to achieve their intended purpose | |
| MDS 1009 | Devices incorporating software / utilising software / controlled by software, including devices intended for controlling, monitoring or directly influencing the performance of active or active implantable devices | ||
| MDS 1010 | Devices with a measuring function | ||
| MDS 1011 | Devices in systems or procedure packs | ||
| MDS 1012 | Products without an intended medical purpose listed in Annex XVI to Regulation (EU) 2017/745 | ||
| MDS 1013 | Class III custom-made implantable devices | ||
| MDS 1014 | Devices incorporating as an integral part an in vitro diagnostic device |
| MDT CODE | Devices for which specific technologies or processes are used | Examples of device manufacturing technologies |
|---|---|---|
| MDT 2001 | Devices manufactured using metal processing | 3 d printing, Casting, Welding 3d printing (metal), turning (metal), anodization, passivation, polishing, surface modification, laser tube cutting, honing |
| MDT 2002 | Devices manufactured using plastic processing | Injection molding, Extrusion, Bonding polymer compounding, 3d printing (plastics), thermoforming, blow moulding, turning (plastics) |
| MDT 2003 | Devices manufactured using non-metal mineral processing (e.g. glass, ceramics) | Ceramic sintering, ceramic compounding, |
| MDT 2004 | Devices manufactured using non-metal non-mineral processing (e.g. textiles, rubber, leather, paper) | Weaving, knitting |
| MDT 2005 | Devices manufactured using biotechnology | Fermentation using cell cultures, enzymatic production processes, purification and modification of biomolecules |
| MDT 2006 | Devices manufactured using chemical processing | Compounding, buffering |
| MDT 2007 | Devices which require knowledge regarding the production of pharmaceuticals | production, handling and incorporation into a device of substances which, if used separately, can be considered to be a medicinal product |
| MDT 2008 | Devices manufactured in clean rooms and associated controlled environments | |
| MDT 2009 | Devices manufactured using processing of materials of human, animal, or microbial origin | Handling, dissection, storage, processing, inactivation and sterilization of human and animal tissues |
| MDT 2010 | Devices manufactured using electronic components including communication | |
| MDT 2011 | Devices which require packaging, including labelling | |
| MDT 2012 | Devices which require installation, refurbishment | Installation of devices at the point of use by or under the manufacturer’s responsibility |
| MDT 2013 | Devices which have undergone reprocessing |
Footnotes
(1): This description does NOT refer to procedure packs or systems according to MDR Article 2 (10) and (11) since those are combinations of individual CE-marked products that have been subjected to separate conformity assessments.
(2): The assessment also should consider Section 5.1 of Annex IX applies as well as Validation of SSCP acc. Article 32.
(3): Directive 2006/42/EC of the European Parliament and of the Council of 17 May 2006 on machinery, and amending Directive 95/16/EC (recast) (OJ L 157 9.6.2006, p. 24).
